Press report
Climate change and risksPinewood nematode: an overview in 8 questions and answers
FAQ - The first detection of the pinewood nematode in France, in the Landes forest in November 2025, marks a significant development. Although it had not previously been found in France, there was a high risk that it would sooner or later reach the country. Here are the key facts in eight questions and answers, based on 15 years of research and modelling work conducted by INRAE on this pest.
Published on 16 March 2026

1. What is the pinewood nematode and what is its impact?
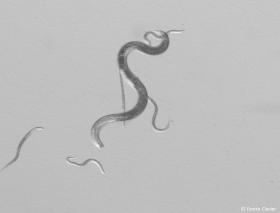
Nematodes are unsegmented roundworms, most of them microscopic. This phylum accounts for a significant share of animal biodiversity. Several species are parasitic and, among them, plant-pathogenic nematodes.
The pinewood nematode (Bursaphelenchus xylophilus) causes pine wilt disease. This microscopic worm (less than 1 mm long) infects several species of conifers, particularly pines. It is transmitted by an insect vector (see Question 3). When the insect feeds on the tree, nematodes enter the sap-conducting tissues. Once inside the tree, they multiply and block sap flow, which leads to the characteristic symptoms of the disease: needles first turn red and then fall, followed by wilting and dieback. Affected trees may die within a few weeks or months, depending on the susceptibility of the host tree and environmental conditions, particularly summer temperatures.
The pinewood nematode is native to North America. First introduced into Japan in the early twentieth century, it has been responsible for the death of millions of pine trees worldwide (see Question 5).
Its introduction and spread in Europe have had major economic, environmental and social consequences. For this reason, B. xylophilus is classified as a 'priority quarantine pest' under European legislation and is therefore subject to specific monitoring and control measures within the European Union.
INRAE scientists have developed molecular markers to genotype populations of B. xylophilus and identify the origin of European populations. Projects are currently under way to reconstruct the evolutionary history of the species and describe the demographic processes affecting populations during invasion. Studying the biology and evolution of the nematode is essential to improve monitoring and control methods (Mallez S et al. 2015).
2. Is it dangerous for humans or animals?
The pinewood nematode attacks only trees and poses no risk to human or animal health.
3. How does it spread from tree to tree?

The pinewood nematode cannot move from one tree to another on its own. It is transmitted by an insect vector, the beetle Monochamus galloprovincialis. This insect becomes a carrier of the nematode when its larvae develop in an infected tree. In spring, Monochamus beetles emerge from dead branches or sections of trunk. The insects undergo a two- to three-week period of sexual maturation. During this period, they feed on the green shoots of healthy trees. Around 75% of tree infections by B. xylophilus occur during this period. This first transmission phase lasts around 10 weeks, as the beetles continue feeding after the sexual maturation phase.
At the end of the season, the beetles select declining pines as egg-laying sites. They may then transmit the nematode to these trees (in around 25% of cases). If the selected trees are already infected by the nematode, it can colonise the eggs, giving rise to a new generation of infected insect vectors.
Research conducted by INRAE shows that the insect vector travels an average of 2 km a day in a homogeneous pine forest and can cover distances of up to 9 km during the transmission phase, which lasts around 10 weeks (Robinet et al 2019).
4. How does the pinewood nematode spread over long distances?
Trade, particularly in wood and bark, is responsible for its long-distance spread. Larvae of the insect vector carrying the nematode may still be present in transported plant material, including the wood used as packaging or pallets. Adult beetles also exhibit hitch-hiking behaviour that can allow them to travel long distances when they are on transported wood.
5. Where does it originate from and where has it been detected?
The pinewood nematode is native to North America, where it does not cause major damage because North American pines are resistant or tolerant after a long period of co-evolution with the nematode. By contrast, it causes considerable damage in regions where it has been introduced. This is the case in Asia: in Japan (detected in 1905), China (detected in 1982), South Korea (detected in 1988) and Taiwan (detected in 1995). It rapidly spread throughout the continent and has been responsible for the mortality of several million trees in Japan and China.
It was first detected in Europe in 1999 in Portugal, then in Spain (2008), Armenia (2022) and now in France (2025, in Seignosse in the Landes region) (Mallez S. et al. 2021).
The Landes forest is highly homogeneous and mainly composed of maritime pine (75%), a host species with a high susceptibility rating (category 5). The mild climate and the high abundance of the insect vector create an environment conducive to its spread.
6. What can be done once the pinewood nematode has been detected?
If the pinewood nematode is detected, European Union legislation provides for mandatory management measures to limit the risk of spread of the nematode and its insect vector. These include clear-cutting within a radius of 100 to 500 metres around infested trees, depending on local conditions at the outbreak site. Other measures are also implemented within a 20 km radius of the outbreak, such as a ban on logging and the transport of wood, combined with active monitoring of declining trees.
Based on work carried out with Portuguese colleagues, INRAE scientists have shown that clear-cutting may fail to prevent spread (Robinet Ch. et al. 2020).
The published results suggest that targeted and early management, involving the selective felling of infected trees, may be preferable. However, the difficulty lies in detecting these trees, as infected trees may remain asymptomatic for long periods or go unnoticed within forest stands. Research is currently under way to improve detection using remote sensing combined with AI-assisted image analysis. At the European level, the EU has asked the European Food Safety Authority (EFSA) to assess these management measures, drawing on INRAE’s work as a reference.
7. What long-term control and prevention measures are available for the pinewood nematode?

Studies are currently under way to identify maritime pine varieties that are resistant or tolerant to the pinewood nematode, as well as the associated genetic factors. Tree populations and improved varieties already present in the Landes forest, different provenances from the natural range of maritime pine (Morocco, Spain, Portugal and Corsica), and hybrid varieties (notably Landes × Corsica) are currently being evaluated.
The tests are carried out in a containment greenhouse and in biosafety level 2 laboratories and technical facilities within the EMERGREEN plateform (INRAE Nouvelle Aquitaine Centre Bordeaux), which has been authorised by the prefecture to handle the pinewood nematode since late February 2025.
Results and genetic gains obtained in Japan on several pine species have been encouraging. Spain has also identified and selected pine families that are more resistant. Developing resistant varieties must be an integral part of pinewood nematode prevention, although this measure alone will not be sufficient (Robinet, C., Raffin, A., Jactel, H. et al. 2024).
Ongoing studies, particularly at INRAE, also show that diversifying the landscape of maritime pine plantations could help slow the spread of the disease:
- through the planting of non-host species such as stone pine (Pinus pinea) (van Halder I. et al. 2022)
- through the presence of broadleaved species (Nunes P. et al. 2021)
8. How is research organised at the national and international level?
INRAE teams are working across several complementary areas to combat the pinewood nematode. These include improving methods for early detection and monitoring, and developing new ones, including remote sensing and insect trapping networks; studying the genetic resistance of maritime pine in order to develop resistant varieties, advancing knowledge of the biology and evolution of the nematode and its insect vector; and developing silvicultural control methods through the diversification of tree species and landscape management (Robinet et al. 2024). In this area, several European projects have already produced recommendations on forest management practices and tree improvement to support more resilient forests (HOMED, B4EST). A new project now under way, FORSAID, involving 10 EU countries, is exploring innovations to improve the management of forest pests, including the pinewood nematode. A major scientific assessment and several collaborations with China, Spain and Portugal are also under way on tree species diversification and landscape management.
Bibliographical References
Mallez, S., Castagnone, C., Espada, M. et al. (2015). Worldwide invasion routes of the pinewood nematode: What can we infer from population genetics analyses? Biological Invasions, 17(4), 1199-1213. https://doi.org/10.1007/s10530-014-0788-9 / hal-02638088v1
Robinet et al. (2019). Modeling the distances traveled by flying insects based on the combination of flight mill and mark-release-recapture experiments. Ecological Modelling DOI : 0.1016/j.ecolmodel.2019.04.006 / hal-02620601
Mallez S., Castagnone C., Lombaert E et al. (2021). Inference of the worldwide invasion routes of the pinewood nematode Bursaphelenchus xylophilus using approximate Bayesian computation analysis. Peer Community Journal, 1: e56. https://doi.org/10.24072/pcjournal.64 / hal-03515563v1
Nunes P. et al. (2021). Modelling Monochamus galloprovincialis dispersal trajectories across a heterogeneous landscape to optimize monitoring by trapping networks. Landscape ecology 36, 931–94. https://doi.org/10.1007/s10980-020-01188-1 / hal-03610281v1 )
Robinet Ch. et al. (2020). Effectiveness of clear‐cuttings in non‐fragmented pine forests in relation to EU regulations for the eradication of the pine wood nematode. Journal of Applied Ecology, https://doi.org/10.1111/1365-2664.13564
Robinet, C., Raffin, A., Jactel, H. et al. (2024). Improving monitoring and management methods is of the utmost importance in countries at risk of invasion by the pinewood nematode. Annals of Forest Science, 81, 16, https://doi.org/10.1186/s13595-024-01233-y
van Halder I. et al. (2022). Pinus pinea: a natural barrier for the insect vector of the pine wood nematode? Anal of forest science, 10.1186/s13595-022-01159-3 / hal-03820711
Nunes P. et al. (2021). Modelling Monochamus galloprovincialis dispersal trajectories across a heterogeneous landscape to optimize monitoring by trapping networks. Landscape ecology, 36, 931-94, 10.1007/s10980-020-01188-1
Robinet, C., Raffin, A., Jactel, H. et al. (2024). Improving monitoring and management methods is of the utmost importance in countries at risk of invasion by the pinewood nematode. Annals of Forest Science, 81, 16, 10.1186/s13595-024-01233-y


