Bioeconomy Reading time 3 min
Extracting rare-earth elements from plants
Published on 04 May 2020

What are the challenges regarding rare-earth elements?
Jean Louis Morel: “Rare-earth” elements such as cerium, indium, neodymium etc. (1) are crucial to several industry sectors—automotive, aeronautics, new technologies—since they can be found in permanent magnets, flat screens, smart phones, batteries, wind turbines, and solar panels. 90% of the known global reserves of these rare-earth metals—which are not that rare after all—are in China and they represent an extremely important strategic economic issue. Certain methods of rare-earth metal extraction are not appropriate from the environmental perspective, since they consist of excavating matter containing rare-earth metals, placing it in a plastic film, and leaching it with ammonium salts—following the principle of ion exchange. This results in the stripping of soils, which increases risks of erosion and water loss.
These methods are currently being replaced and, in order to restore the degraded soils, plants are being used to provide cover and restore ecosystems. Some of these plants have the particularity of extracting and accumulating rare-earth elements in their leaves, which can then be treated to obtain the elements through the process of hydrometallurgy. This solution, known by the generic name of “agromining,” offers two advantages: avoiding soil erosion and water pollution, and producing useful biomass from which residual rare-earth metals can be extracted. It is crucial to use the right plants and the proper rare-earth extraction procedures according to the plants selected.
What is the current state of research on rare-earth agromining?
J-L M. : We have identified a rare-earth hyperaccumulator plant, a fern called Dicranopteris linearis (2). This plant is capable of accumulating 0.2 to 0.3% (in dry matter weight) of rare-earth metals in their fronds (3). As for the extraction of rare-earth elements using this plant, several procedures have been developed and their viability is currently being assessed at a pilot stage (4). Recently, fundamental research studies on plant physiology have allowed us to move ahead, by identifying how, and in which organs of the plant, rare-earth metals are accumulated (5).
What have you discovered?
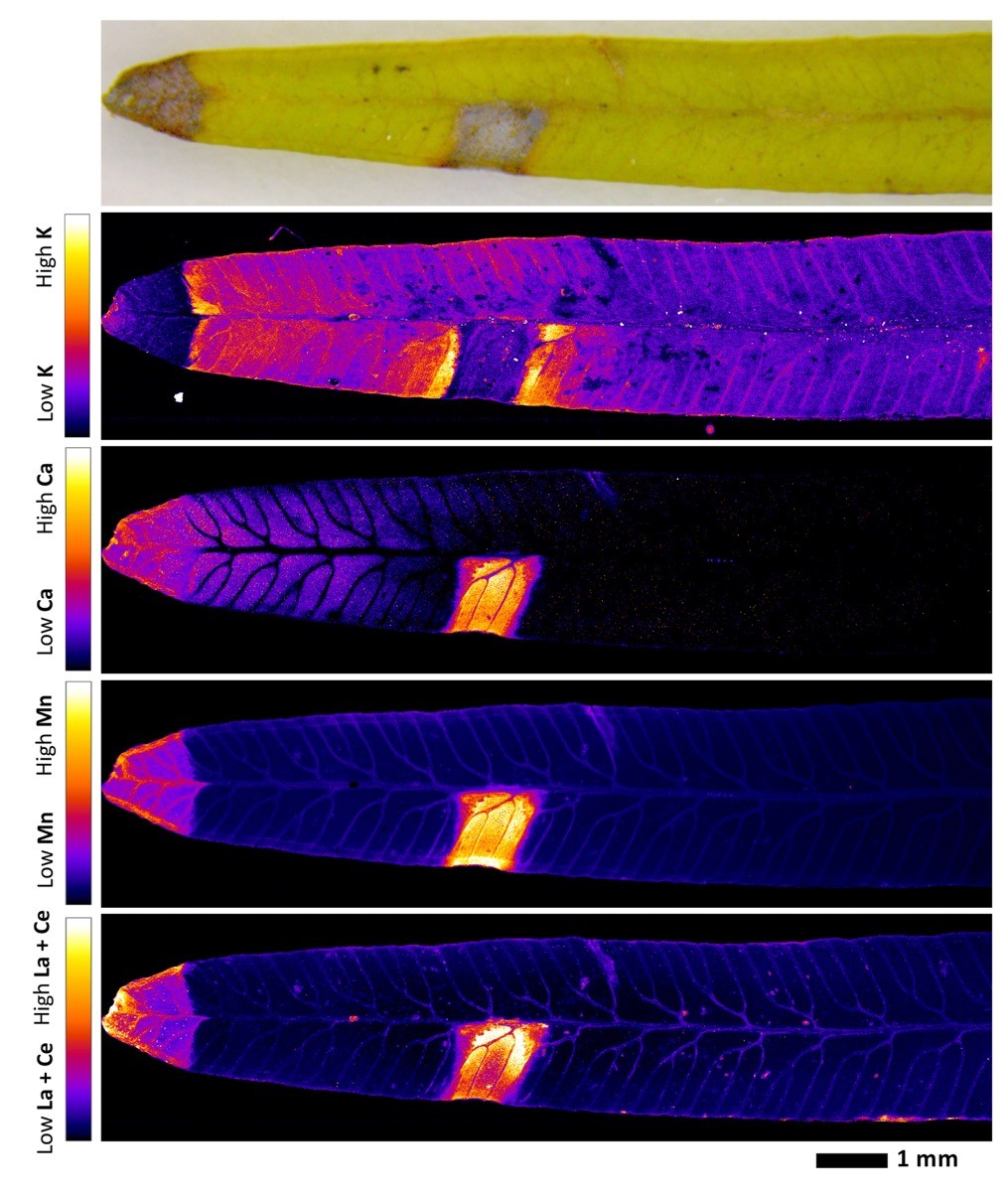
J-L M. : The main discovery is that rare-earth elements accumulate in the necrotic lesions of the leaves—up to 0.5% in dry weight. We have proven this in the case of two rare-earth elements, lanthanum (La) and cerium (Ce), by using a synchrotron, which allows us to visualise the elements directly in the plant’s organs. In fact, we observed that lanthanum and cerium are accumulated in the same areas as manganese, and this accumulation produces necrotic lesions. Our hypothesis is that, in the necrotic lesions, cellular disorganisation accelerates evapotranspiration (6), causing a suction pump effect. Elements such as La, Ce, and aluminium—another toxic metal chemically similar to La and Ce—are passively accumulated in this “waste” area. The area that contains toxic elements is sacrificed to save other cells. We also observed that all these metals are located in the same area as silicon (Si), an element that serves to stabilise them, thus contributing to the detoxifying process.
We now have a better understanding of the mechanisms of rare-earth element accumulation in Dicranpteris linearis, which differ from those of plants that accumulate metals like nickel and zinc by stocking them in their aerial organs (stem, leaves and flowers).
What's next?
J-L M. : As part of our agreement with the Universities of Sun Yat-sen and Queensland (7), we will assess other accumulating plants in terms of their silicon content, which could work as a marker. We have around 20 candidate plants. We will also continue working on optimizing the extraction procedures before expanding the project. Finally, we will conduct life-cycle and economic assessments to evaluate the viability of the processes.
(1) "Rare earths" denote a set of 17 metals of the periodic table called "lanthanides", which share similar properties, to which scandium (Sc) and yttrium (Y) are added.
(2) Collaboration with the University Sun Yat-sen in Canton, China, which led to the creation of the ECOLAND LIA by INRAE and the University of Lorraine.
(3) Wenshen Liu’s PhD dissertation, 2019.
(4) The Reactions and Chemical Engineering Laboratory (LRGP) joint unit of the University of Lorraine, the CNRS, Marie-Odile Simonnot’s team.
(5) Collaboration with the University of Queensland, Australia.
(6) In plants, water rises from the roots to the leaves via very thin vessels. The upward movement of these water columns is caused by evapotranspiration (loss of water in the leaves via the stomata) which creates a passive suction effect from the bottom up.
(7) Collaboration within the framework of the ECOLAND LIA, and the joint laboratory Sucre, between the Universities of Lorraine and Queensland.
Liu et al. 2020. Spatially-resolved localization of lanthanum and cerium in the rare earth element hyperaccumulator fern Dicranopteris linearis from China. Environmental Science and Technology. DOI: 10.1021/acs.est.9b05728
