Food, Global Health Reading time 2 min
AvBD11, the small protein found naturally in eggs that is packed with therapeutic potential
Published on 10 January 2020

The chicken egg contains about a hundred of molecules potentially involved in egg natural defense. These include antimicrobial peptides of the β-defensin family, such as the « avian β-defensin 11 » (AvBD11), which is uniquely found in the egg. A study previously published by one of the consortium partners demonstrated that this protein exhibits a broad antibacterial spectrum (4). Preliminary analyses of the protein sequence pointed out the atypical size of AvBD11 and the potential presence of two β-defensin-like domains (5), whereas all other avian β-defensins contains only one single domain.
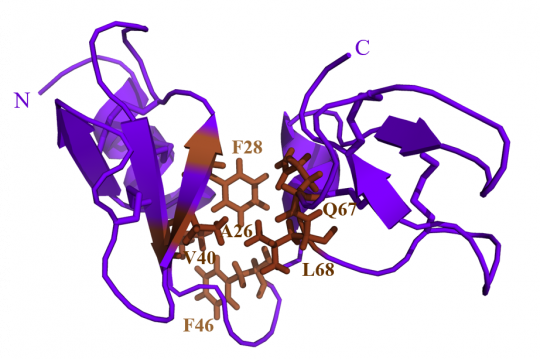
The structural characterization of egg-purified AvBD11 by nuclear magnetic resonance (NMR) reveals that this protein has a compact structure consisting of two interacting β-defensin domains. This unique structural feature never described before, is proposed as the archetype of a new structural family called « avian double-β-defensins (Av-DBD) ». The two corresponding domains were individually obtained by chemical synthesis and their activities tested, alone or in synergy, to assess their respective contribution in the biological activity of native AvBD11. The results demonstrate that the antibacterial, antiparasitic and anti-invasive activities mainly rely on the first domain, while antiviral and cytotoxic activities are only observed for the whole protein, which underlines the pivotal and complementarity role of both domains for certain biological activities.
AvBD11 is a protein with multiple activities that are essential for the embryonic development of the bird, although it likely possesses other yet unknown functions. Besides its interest in developmental and evolutionary biology, such biological activities exhibited by AvBD11, could potentially be used in human health for the development of polyvalent therapeutic agents targeting infectious diseases but also cell disorders associated with cancer (patent application pending). This work is one example of getting inspired by natural molecules that have been shaped during evolution and “time-felt” to protect the avian embryo, to develop new efficient therapeuticals.
1 This work was conducted as part of two research projects financed by Région Centre-Val de Loire: « MUSE » (Medicinal USe of Eggs », 2014-00094512) coordinated by S. Réhault-Godbert, and « SAPhyR-11 » (Structure/Activity and PHYlogenetic Relationships of AvBD11, 2017-119983) coordinated by N. Guyot. The MUSE project was dealing with structural and functional characterization of peptides and proteins purified from egg and with the assessment of their potential value in human and animal Health. This project allowed the identification of a promising peptide, AvBD11, which was further characterized in the SAPhyR-11 project.
2 Proteinaceous layer surrounding the egg yolk
3 Inhibitory activity of cell migration (within a matrix substrate) allowing a control of the dissemination of cells within a tissue or an organism
4 Purification and characterization of avian beta-defensin 11, an antimicrobial peptide of the hen egg. Hervé-Grépinet et al. Antimicrob Agents Chemother doi: 10.1128/AAC.00204-10.
5 Part of a protein forming a basic structural and functional unit
|
References Structure, function and evolution of Gga-AvBD11, the archetype of a new structural avian-double-β-defensin family, Guyot et al., Proceedings of the National Academy of Sciences, doi: 10.1073/pnas.1912941117 Demande de brevet FR18 57411 du 9/08/2018 et PCT/EP2019/071388 « PEPTIDES DERIVES D'UNE BETA-DEFENSINE 11 AVIAIRE ET LEURS UTILISATIONS » |
