Food, Global Health Reading time 2 min
The origin of satiety: brain cells that change shape after a meal
Published on 03 March 2020
The neuronal circuits in our brain governing feelings of hunger and satiety can modify their connections, thereby adjusting feeding behaviour to living conditions and maintaining a balance between food intake and calorie expenditure. Scientists suspect that this plasticity could be altered for obese subjects.
In a new study conducted on mice, a team led by Alexandre Benani, a CNRS researcher at the Centre for Taste and Feeding Behavior (CNRS/INRAE/University of Burgundy/AgroSup Dijon), has shown that these circuits are activated on the time scale of a meal, subsequently regulating feeding behaviour. However, this activation does not occur through a change in the circuit’s “connections.”
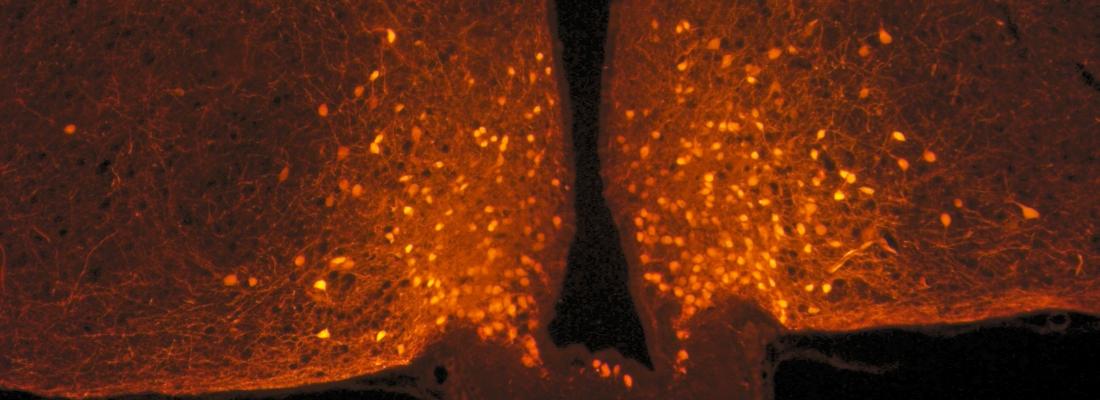
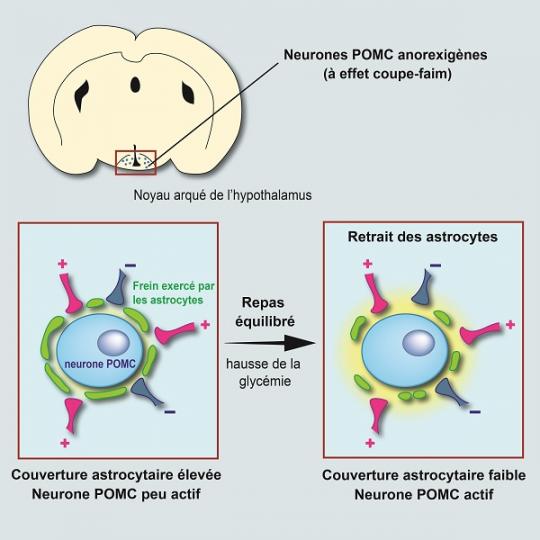
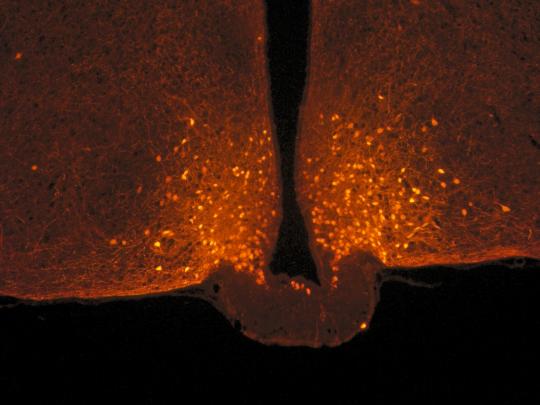
Scientists focused on POMC neurons in the hypothalamus, located at the base of the brain, which are known for limiting food intake. They are connected to a large number of neurons from other parts of the brain, with the connections of this circuit being malleable: they can be made and unmade very quickly based on hormonal fluctuations. Researchers observed that this neuronal circuit is not modified after a balanced meal, but that other nerve cells associated with POMC neurons, known as astrocytes, actually change form.
Astrocytes are star-shaped nerve cells that were first studied for their supporting role with respect to neurons. Under usual conditions, they sheathe POMC neurons and act somewhat like brake pads by limiting their activity. After a meal, blood glucose levels (glycaemia) temporarily increase, with astrocytes detecting this signal and retracting in less than one hour: once this “brake” is released, POMC neurons are activated, ultimately promoting the feeling of satiety.
Surprisingly, a meal that is high in fats does not lead to this remodelling. Does this mean that lipids are less effective in satisfying hunger? The scientists are trying to determine whether they trigger satiety through another circuit. It also remains to be seen whether sweeteners have the same effects, or whether they lure the brain by providing an addictive sensation of sweetness without satisfying hunger.
* The study was led by the Centre for Taste and Feeding Behavior (CNRS/Inrae/University of Burgundy/Agrosup Dijon), in close collaboration with colleagues from the Institute of Molecular and Cellular Pharmacology (CNRS/Université Côte d’Azur), Institute for Functional Genomics (CNRS/Inserm/ University of Montpellier), and University of Luxembourg, along with contributions from l’Unité de biologie fonctionnelle et adaptative (CNRS/Université de Paris) and Institute of Psychiatry and Neuroscience of Paris (Inserm/Université de Paris).
|
|
|
Remodelling of the satiety circuit of POMC neurons after a balanced meal. Red box: area corresponding to the photo at right. © Alexandre Benani / CNRS / CSGA |
POMC neurons (orange dots) in the hypothalamus of a mouse, located at the base of the brain. Photo taken from a mouse using a confocal microscope. © Danaé Nuzzaci / CNRS / CSGA |
|
Reference Postprandial hyperglycemia stimulates neuroglial plasticity in hypothalamic POMC neurons after a balanced meal, Danaé Nuzzaci, Céline Cansell, Fabienne Liénard, Emmanuelle Nédélec, Selma Ben Fradj, Julien Castel, Ewout Foppen, Raphael Denis, Dominique Grouselle, Amélie Laderrière, Aleth Lemoine, Alexia Mathou, Virginie Tolle, Tony Heurtaux, Xavier Fioramonti, Etienne Audinat, Luc Pénicaud, Jean-Louis Nahon, Carole Rovère, Alexandre Benani, Cell Reports, le 3 March 2020. DOI : 10.1016/j.celrep.2020.02.029 |
